Guangzhou LBP Medicine Science & Technology Co., Ltd. has reached a significant milestone in its global growth strategy with the successful attainment of CE-IVDR certification for 14 pathology diagnostic products. This accomplishment represents a crucial step in elevating the international presence and credibility of China's medical diagnostics industry.
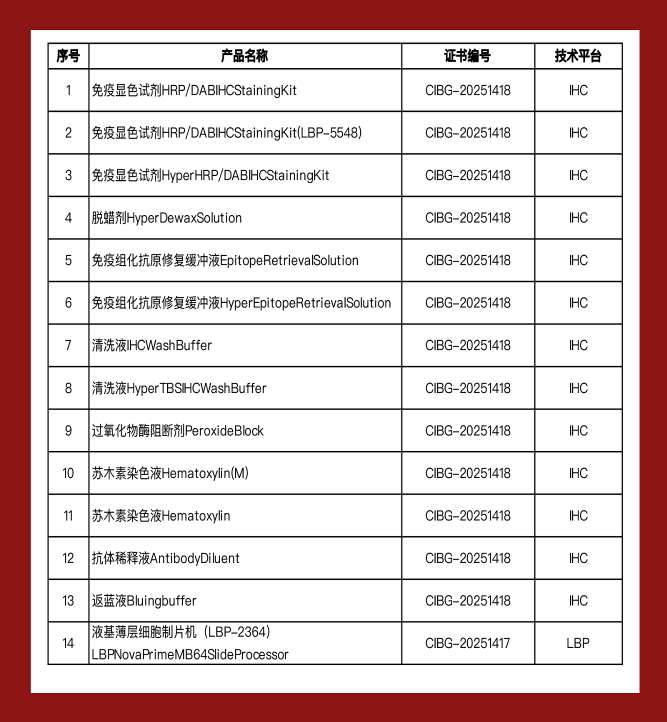
The certified portfolio spans two major categories: liquid-based cytology (LBC) and immunohistochemistry (IHC). At the forefront is the LBP-2364, a fully automated thin-layer cytology processor independently developed by the company. As a flagship product, it symbolizes China's progress in LBC technology and showcases LBP’s innovation on the global stage.
In addition, 13 IHC-related products, including secondary antibodies and auxiliary reagents, have also received CE-IVDR certification. These reagents are fully compatible with the Hyper S9 Fully Automated IHC Staining System, which was previously certified, creating a cohesive and efficient end-to-end solution—from sample preparation to staining—for modern pathology laboratories.
This certification milestone marks a notable shift in the global pathology landscape, which has long been dominated by imported technologies. LBP’s recognition through CE-IVDR affirms the growing competitiveness of Chinese-developed solutions and underscores the company’s role as a leader in high-quality, accessible diagnostics.
Guided by China’s Belt and Road Initiative, LBP has steadily expanded its international business presence across key markets, including Singapore, Japan, Russia, Turkey, Spain, Ecuador, Vietnam, Indonesia, Slovakia, Malaysia, Saudi Arabia, Egypt, Iran, Myanmar, Ukraine, and the Congo.This expanding global reach reflects the company’s commitment to advancing the efficiency, precision, and accessibility of tumor diagnostics worldwide.